Chapter 1: Q.13E (page 9)
Find the potentials of the following electrochemical cell:
\(Cd|C{d^{2 + }},M = 0.10||N{i^{2 + }},M = 0.50|Ni\)
Short Answer
The potentials of the following electrochemical cell is \({E_{cell}} = 0.167V\)
Learning Materials
Features
Discover
Chapter 1: Q.13E (page 9)
Find the potentials of the following electrochemical cell:
\(Cd|C{d^{2 + }},M = 0.10||N{i^{2 + }},M = 0.50|Ni\)
The potentials of the following electrochemical cell is \({E_{cell}} = 0.167V\)
All the tools & learning materials you need for study success - in one app.
Get started for free
Which molecule has a molecular mass of 28.05 amu?
(a) \({\rm{HC}} \equiv {\rm{CH}}\)
(b)
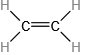
(c)
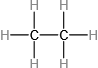
Write conversion factors (as ratios) for the number of:
(a) kilometers in 1 mile
(b) liters in 1 cubic foot
(c) grams in 1 ounce
Question:The gas tank of a certain luxury automobile holds 22.3 gallons according to the owner’s manual. If the densityof gasoline is 0.8206 g/mL, determine the mass in kilograms and pounds of the fuel in a full tank.
Does the standard enthalpy of formation of \({{\bf{H}}_{\bf{2}}}{\bf{O}}\left( {\bf{g}} \right)\)differ from ΔH° for the reaction \({\bf{2}}{{\bf{H}}_{\bf{2}}}\left( {\bf{g}} \right){\bf{ + }}{{\bf{O}}_{\bf{2}}}\left( {\bf{g}} \right) \to {\bf{2}}{{\bf{H}}_{\bf{2}}}{\bf{O}}\left( {\bf{g}} \right)\)?
A large piece of jewelry has a mass of 132.6g. A graduated cylinder initially contains 48.6 mL water. When the jewellery is submerged in the graduated cylinder, the total volume increases to 61.2 mL.
a) Determine the density of this piece of jewelry.
b) Assuming that the jewellery is made from only one substance, what substance is it likely to be? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.