Chapter 1: Q58 E (page 66)
Write conversion factors (as ratios) for the number of:
(a) kilometers in 1 mile
(b) liters in 1 cubic foot
(c) grams in 1 ounce
Short Answer
(a) 1.6093 km/mile
(b) 28.317 L/\(f{t^3}\)
(c) 31.103 gm/oz
Learning Materials
Features
Discover
Chapter 1: Q58 E (page 66)
Write conversion factors (as ratios) for the number of:
(a) kilometers in 1 mile
(b) liters in 1 cubic foot
(c) grams in 1 ounce
(a) 1.6093 km/mile
(b) 28.317 L/\(f{t^3}\)
(c) 31.103 gm/oz
All the tools & learning materials you need for study success - in one app.
Get started for free
A large piece of jewelry has a mass of 132.6g. A graduated cylinder initially contains 48.6 mL water. When the jewellery is submerged in the graduated cylinder, the total volume increases to 61.2 mL.
a) Determine the density of this piece of jewelry.
b) Assuming that the jewellery is made from only one substance, what substance is it likely to be? Explain.
What are the allowed values for each of the four quantum numbers:n,l,ml, andms?
Round off each of the following numbers to two significant figures:
(a) 0.436
(b) 9.000
(c) 27.2
(d) 135
(e) 1.497 × 10-3
(f) 0.445
Which molecule has a molecular mass of 28.05 amu?
(a) \({\rm{HC}} \equiv {\rm{CH}}\)
(b)
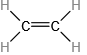
(c)
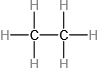
Classify the six underlined properties in the following paragraph as chemical or physical:
Fluorine is a pale yellowgas that reacts with most substances. The free elementmelts at -2000C and boils at -188 °C. Finely divided metals burn in fluorine with a bright flame. Nineteen grams of fluorine will react with 1.0 gram of hydrogen.
What do you think about this solution?
We value your feedback to improve our textbook solutions.