Chapter 3: Q67 E (page 172)
What volume of a 0.20-M K2SO4 solution contains 57 g of K2SO4?
Short Answer
1.6 L of 0.20 M K2SO4solutioncontains 57 g of K2SO4.
Learning Materials
Features
Discover
Chapter 3: Q67 E (page 172)
What volume of a 0.20-M K2SO4 solution contains 57 g of K2SO4?
1.6 L of 0.20 M K2SO4solutioncontains 57 g of K2SO4.
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the molecular mass of the following compounds:
(a)
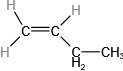
(b)
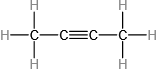
(c)
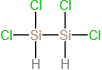
(d)
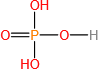
Calculate the molar mass of each of the following compounds:
(a) hydrogen fluoride, HF
(b) ammonia, NH3
(c) nitric acid, HNO3
(d) silver sulfate, Ag2SO4
(e) boric acid, B(OH)3
Question-Determine the empirical formulas for the compounds with the following percentage composition:
(a) 43.6% phosphorous and 56.4% oxygen
(b) 28.7%K, 1.5%H, 22.8%P and 47%O
Question: Determine the following to four significant figures-
(a) The percent composition of hydrazoic acid, HN3
(b) The percent composition of TNT, C6H2CH3(NO2)3
(c) The percent of SO42- in Al2(SO4)3
Calculate the molar mass of each of the following:
(a) S8
(b) C5H12
(c) Sc2(SO4)3
(d) CH3COCH3 (acetone)
(e) C6H12O6(glucose)
What do you think about this solution?
We value your feedback to improve our textbook solutions.