Chapter 3: Q.5E (page 166)
Determine the molecular mass of the following compounds:
(a)
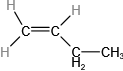
(b)
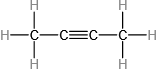
(c)
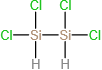
(d)
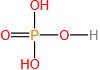
Short Answer
The total molecular or formula mass of each compound will be:
(a)56 amu
(b)54 amu
(c)200 amu
(d)97 amu
Learning Materials
Features
Discover
Chapter 3: Q.5E (page 166)
Determine the molecular mass of the following compounds:
(a)

(b)

(c)

(d)

The total molecular or formula mass of each compound will be:
(a)56 amu
(b)54 amu
(c)200 amu
(d)97 amu
All the tools & learning materials you need for study success - in one app.
Get started for free
A 55-kg woman has 7.5 × 10−3 mol of hemoglobin (molar mass = 64,456 g/mol) in her blood. How many hemoglobin molecules is this? What is this quantity in grams?
Question- Determine the empirical formula for the compounds with the following percentage composition:
(a) 15.8% carbon and 84.2% sulfur.
(b) 40% carbon, 6.7% hydrogen, and 53.3% oxygen.
A 50.0-g sample of industrial wastewater was determined to contain 0.48 mg of mercury. Express the mercury concentration of the wastewater in ppm and ppb units.
What is the molecular formula of a compound with a percent composition of 49.47% C, 5.201% H, 28.84% N, 16.48% O, and a molecular mass of 194.2 amu?
What volume of a 1.50-MKBr solution contains 66.0 g KBr?
What do you think about this solution?
We value your feedback to improve our textbook solutions.