Chapter 2: Q52E (page 126)
Question : Name the following compounds:
- NaF
- Rb2O
- BCl3
- H2Se
- P4O6
- ICl3
Short Answer
- Sodium fluoride
- Rubidium oxide
- Boron trichloride
- Hydrogen selenide
- Tetra phosphorus hexoxide
- Iodine trichloride
Learning Materials
Features
Discover
Chapter 2: Q52E (page 126)
Question : Name the following compounds:
All the tools & learning materials you need for study success - in one app.
Get started for free
Open theBuild an Atom simulation (http://openstaxcollege.org/l/16PhetAtomBld)and click on the
Atom icon.
a. Pick any one of the first 10 elements that you would like to build and state its symbol.
b. Drag protons, neutrons, and electrons onto the atom template to make an atom of your element.
State the numbers of protons, neutrons, and electrons in your atom, as well as the net charge and mass number.
c. Click on “Net Charge” and “Mass Number,” check your answers to (b), and correct, if needed.
d. Predict whether your atom will be stable or unstable. State your reasoning.
e. Check the “Stable/Unstable” box. Was your answer to (d) correct? If not, first predict what you can do to make a Stable atom of your element, and then do it and see if it works. Explain your reasoning.
Using the periodic table, identify the heaviest member of each of the following groups:
(a) Alkali metals
(b) Chalcogens
(c) Noble gases
(d) Alkaline earth metals
For each of the following pairs of ions, write the symbol for the formula of the compound they will form:
Write the molecular and empirical formulas of the following compounds:
a)
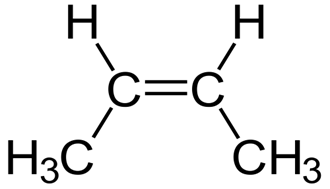
(b)
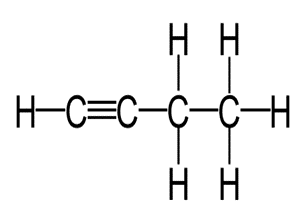
(c)
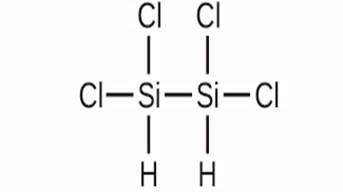
(d)
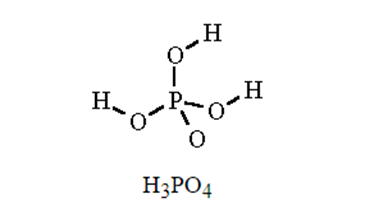
Using the periodic table, classify each of the following elements as a metal or a nonmetal, and then further classify each as a main-group (representative) element, transition metal, or inner transition metal:
What do you think about this solution?
We value your feedback to improve our textbook solutions.