Chapter 2: Q36E (page 128)
Use the build a molecule simulation (http://openstaxcollege.org/1/16molbuilding) to repeat Exercise 2.34, but build a molecule with three carbons, seven hydrogens, and one chlorine.
- Draw the structural formula of this molecule and state its name.
- Can u arrange these atoms to make a different molecule? If so, draw its structural formula and state its name.
- How are molecules drawn in (a) and (b) the same? How do they differs? What are they called (the type of relationship between these molecules, not their names)?
Short Answer
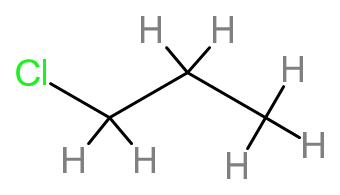
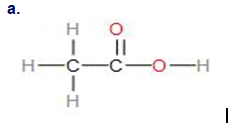
(a) Structural formula:
\(\)
Name- \(1 - chloropropane\)
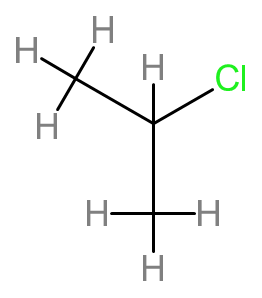
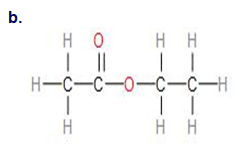
b.
Name- 2- Chloropropane
c. Molecular formula is same, structure is different, structural isomers.