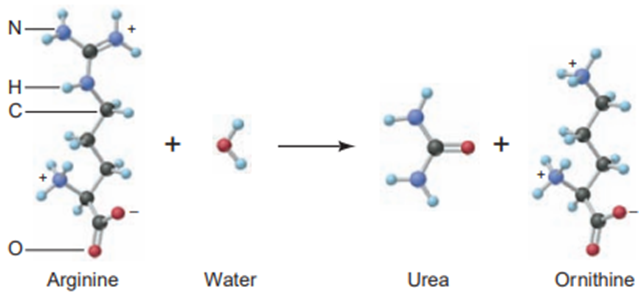
Chapter 3: Q3.27P (page 132)
The effectiveness of a nitrogen fertilizer is determined mainly by its mass % N. Rank the following fertilizer, most effective first: potassium nitrate; ammonium nitrate; ammonium sulfate; urea,
Short Answer
The Effectiveness of the given fertilizers is
Urea > ammonium nitrate > ammonium sulfate > potassium nitrate.