Chapter 3: Q155CP (page 139)
The human body excretes nitrogen in the form of urea, NH2CONH2. The key biochemical step in urea formation is the reaction of water with arginine to produce urea and ornithine:
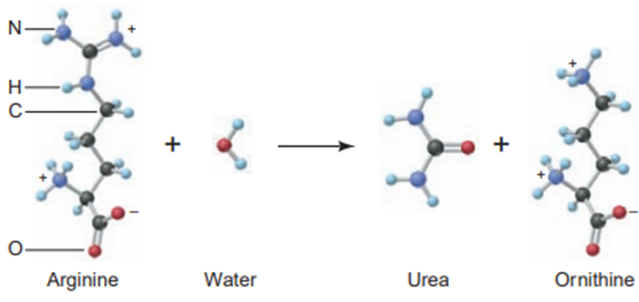
(a) What is the mass percent of nitrogen in urea, arginine, and ornithine? (b) How many grams of nitrogen can be excreted as urea when 135.2 g of ornithine is produced?
Short Answer
- The mass percentage of nitrogen in urea as: 46.65% N in urea, 32.16% N in arginine and 21.2 % in ornithine.
There are 28.66 g Ncan be excreted as urea when 135.2 g or ornithine is produced.