Chapter 3: Q.3.105P (page 89)
How many grams of NaH2PO4 are needed to react with 43.74 mL of 0.285 M NaOH?
Short Answer
The mass of NaH2PO4needed to reactwith 43.74 mL of 0.285 M NaOHis 0.747 g of NaH2PO4.
Learning Materials
Features
Discover
Chapter 3: Q.3.105P (page 89)
How many grams of NaH2PO4 are needed to react with 43.74 mL of 0.285 M NaOH?
The mass of NaH2PO4needed to reactwith 43.74 mL of 0.285 M NaOHis 0.747 g of NaH2PO4.
All the tools & learning materials you need for study success - in one app.
Get started for free
Many metals react with oxygen gas to form the metal oxide. For example, calcium reacts as follows:
You wish to calculate the mass of calcium oxide that can be prepared from 4.20 g of and 2.80 g of
(a) How many moles of CaO can be produced from the given mass of Ca ?
(b) How many moles of CaO can be produced from the given mass of
(c) Which is the limiting reactant?
(d) How many grams of CaO can be produced?
List three ways compositional data may be given in a problem that involves finding an empirical formula.
Write balanced equations for each of the following by inserting the correct coefficients in the blanks:
How does a balanced chemical equation apply the law of conservation of mass?
Each of the following balances weighs the indicated numbers of atoms of two elements:
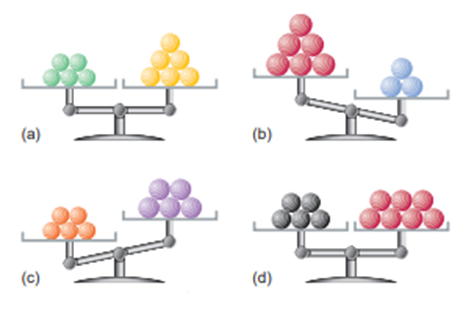
Which element-left, right, or neither,
(a) Has the higher molar mass?
(b) Has more atoms per gram?
(c) Has fewer atoms per gram?
(d) Has more atoms per mole?
What do you think about this solution?
We value your feedback to improve our textbook solutions.