Chapter 3: Q3.30P (page 132)
List three ways compositional data may be given in a problem that involves finding an empirical formula.
Short Answer
The Mass percentage , Mass of components or mass of compound and elements with one unknown.
Learning Materials
Features
Discover
Chapter 3: Q3.30P (page 132)
List three ways compositional data may be given in a problem that involves finding an empirical formula.
The Mass percentage , Mass of components or mass of compound and elements with one unknown.
All the tools & learning materials you need for study success - in one app.
Get started for free
Hemoglobin, a protein in red blood cells, carries from the lungs to the body’s cells. Iron(as ferrous ion, ) makes up 0.33 mass % of hemoglobin. If the molar mass of hemoglobin is 6.8 g/mol, how many ions are inone molecule?
Each of the following balances weighs the indicated numbers of atoms of two elements:
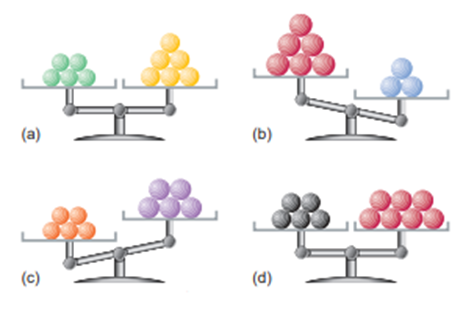
Which element-left, right, or neither,
(a) Has the higher molar mass?
(b) Has more atoms per gram?
(c) Has fewer atoms per gram?
(d) Has more atoms per mole?
The following circles represent a chemical reaction between AB2 and B2:
(a) Write a balanced equation for the reaction. (b) What is the limiting reactant? (c) How many moles of product can be made from 3.0 mol of B2 and 5.0 mol of AB2? (d) How many moles of excess reactant remain after the reaction in part (c)?
Alum [KAI(SO4)2 .xH2O]is used in food preparation, dye fixation, and water purification. To prepare alum, aluminum is reacted with potassium hydroxide and the product with sulfuric acid. Upon cooling, alum crystallizes from the solution. (a) A 0.5404-g sample of alum is heated to drive off the waters of hydration, and the resulting KAI(SO4)2weighs 0.2941 g. Determine the value of and the complete formula of alum. (b) When 0.7500 g of aluminum is used, 8.500 g of alum forms. What is the percent yield?
Chromium(III) oxide reacts with hydrogen sulfide gas to form chromium(III) sulfide and water:
To produce 421 g of
(a) How many moles of are required?
(b) How many grams of are required?
What do you think about this solution?
We value your feedback to improve our textbook solutions.