Chapter 3: Q11P (page 131)
Each of the following balances weighs the indicated numbers of atoms of two elements:
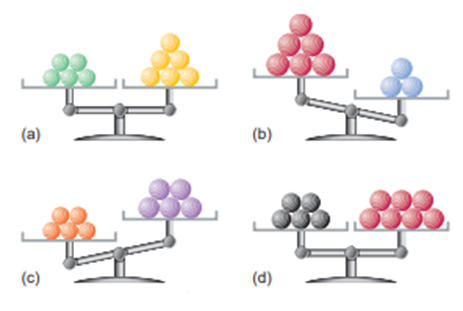
Which element-left, right, or neither,
(a) Has the higher molar mass?
(b) Has more atoms per gram?
(c) Has fewer atoms per gram?
(d) Has more atoms per mole?
Short Answer
The followingbalance weighs the indicated numbers of atoms of two elements
- A-Left; B-Right; C-Left; D-Left
- A-Right; B- Left; C- Right; D- Left
- A-Left; B- Right; C-Left; D-Left
- Avogadro's constantstates that they all have the same number of atoms per mole.


