Chapter 14: Q14.95P (page 623)
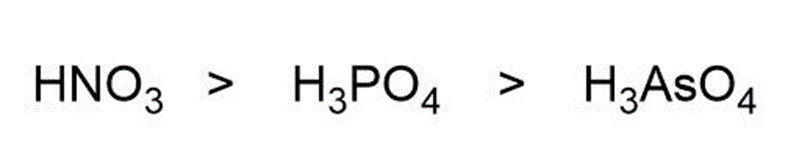
Question: Assuming acid strength relates directly to the electronegativity of the central atom, rank H3PO4,HNO3 , and H3ASO4 in order of increasing acid strength.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 14: Q14.95P (page 623)
Question: Assuming acid strength relates directly to the electronegativity of the central atom, rank H3PO4,HNO3 , and H3ASO4 in order of increasing acid strength.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
One similarity between B and Si is the explosive combustion of their hydrides in air. Write balanced equations for the combustion of androle="math" localid="1653907272447" .
(a) What is the range of oxidation states shown by the elements of Group 5A (15) as you move down the group?
(b) How does this range illustrate the general rule for the range of oxidation states in groups on the right side of the periodic table?
Draw Lewis structures for the following compounds and predict which member of each pair will form hydrogen bonds:
(a) NH3 or AsH3
(b) CH4 or H2O
An important starting material for the manufacture of polyphosphazene is the cyclic molecule (NPCl2)3. The molecule has a symmetrical six-membered ring of alternating N and P atoms, with the Cl atoms bonded to the P atoms. The nitrogen phosphorus bond length is significantly less than that expected for an NP single bond.
(a) Draw a likely Lewis structure for the molecule.
(b) How many lone pairs of electrons do the ring atoms have?
(c) What is the order of the nitrogen-phosphorus bond?
In addition to those in Table 14.3, other less stable nitrogen oxides exist. Draw a Lewis structure for each of the following:
(a) , a dimer of nitrogen monoxide with an bond.
(b) , a dimer of nitrogen monoxide with no bond.
(c) with no bond.
(d) and , products of the ionization of liquid .
What do you think about this solution?
We value your feedback to improve our textbook solutions.