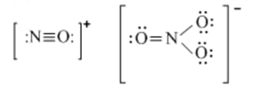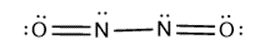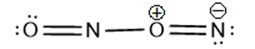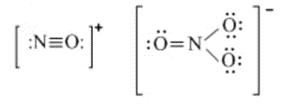Chapter 14: Q105P (page 623)
In addition to those in Table 14.3, other less stable nitrogen oxides exist. Draw a Lewis structure for each of the following:
(a) , a dimer of nitrogen monoxide with an bond.
(b) , a dimer of nitrogen monoxide with no bond.
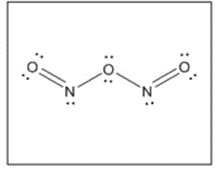
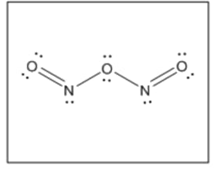
(c) with no bond.
(d) and , products of the ionization of liquid .
Short Answer
The answer is as follow.
(a)
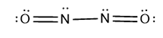
(b)
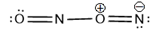
(c)
(d)