Chapter 14: Q102P (page 623)
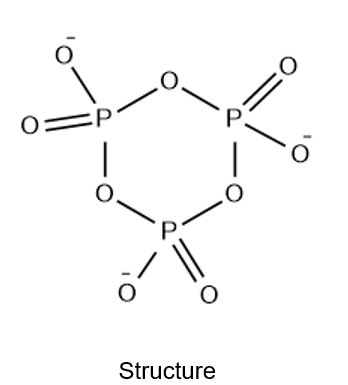
Use the VSEPR model to predict the structure of the cyclic ion
Short Answer
The answer is
Learning Materials
Features
Discover
Chapter 14: Q102P (page 623)
Use the VSEPR model to predict the structure of the cyclic ion
The answer is

All the tools & learning materials you need for study success - in one app.
Get started for free
Bromine monofluoride (BF) disproportionates to bromine gas and bromine tri- and pentafluorides. Use the following to find Δfor the decomposition of BrF to its elements:
Thallium forms the compound . What is the apparent oxidation state of Tl in this compound? Given that the anion is , what is the actual oxidation state of Tl? Draw the shape of the anion, giving its VSEPR class and bond angles. Propose a reason why the compound does not exist as.
Rank the following in order of decreasing
(a) Boiling point: O2, Br2, As(s)
(b): Cl2, Ar, I2
Even though EN values vary relatively little down a group 4A (14), the elements change from nonmetal to metal. Explain.
Which member of each pair has more ioniccharacter in its bonds:
(a) BeF2 or CaF2; (b) PbF2 or PbF4; (c) GeF4 or PF3?
What do you think about this solution?
We value your feedback to improve our textbook solutions.