Chapter 14: Q14.36 P (page 621)
Draw Lewis structures for the following compounds and predict which member of each pair will form hydrogen bonds:
(a) NH3 or AsH3
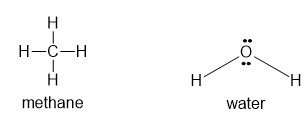
(b) CH4 or H2O
Short Answer
(a) will form hydrogen bonds.
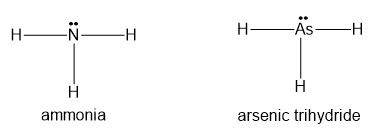
(b) will form hydrogen bonds.
Learning Materials
Features
Discover
Chapter 14: Q14.36 P (page 621)
Draw Lewis structures for the following compounds and predict which member of each pair will form hydrogen bonds:
(a) NH3 or AsH3
(b) CH4 or H2O
(a) will form hydrogen bonds.
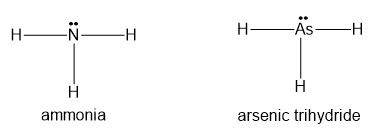
(b) will form hydrogen bonds.

All the tools & learning materials you need for study success - in one app.
Get started for free
Bismuth(V) compounds are such powerful oxidizing agents that they have not been prepared in pure form. How is this fact consistent with the location of Bi in the periodic table?
The electronic transition in Na from 3p1to3s1 gives rise to a bright yellow-orange emission at 589.2 nm. What is the energy of this transition?
Which member of each pair has more covalentcharacter in
its bonds: (a) LiCl or KCl; (b) AlCl3 or PCl3; (c) NCl3 or AsCl3?
Question: (a) What is the range of oxidation states shown by the elements of Group 5A (15) as you move down the group?
(b) How does this range illustrate the general rule for the range of oxidation states in groups on the right side of the periodic table?
Which member of each pair has more ioniccharacter in its bonds:
(a) BeF2 or CaF2; (b) PbF2 or PbF4; (c) GeF4 or PF3?
What do you think about this solution?
We value your feedback to improve our textbook solutions.