Chapter 14: Q14.141CP (page 625)
The electronic transition in Na from 3p1to3s1 gives rise to a bright yellow-orange emission at 589.2 nm. What is the energy of this transition?
Short Answer
The energy of this transition is.
Learning Materials
Features
Discover
Chapter 14: Q14.141CP (page 625)
The electronic transition in Na from 3p1to3s1 gives rise to a bright yellow-orange emission at 589.2 nm. What is the energy of this transition?
The energy of this transition is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Which member of each pair has more ioniccharacter in its bonds:
(a) BeF2 or CaF2; (b) PbF2 or PbF4; (c) GeF4 or PF3?
The interhalogen IF undergoes the reaction depicted below (I is purple and F is green):
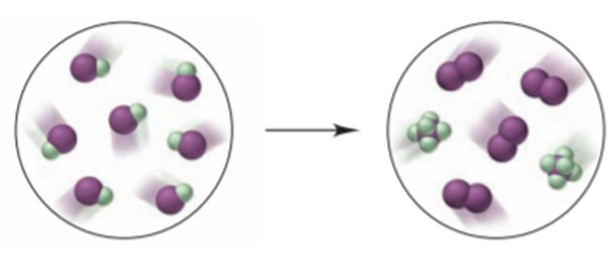
(a) Write the balanced equation.
(b) Name the interhalogen product.
(c) What type of reaction is shown?
(d) If each molecule of IF represents mol, what mass of each product forms?
Bromine monofluoride (BF) disproportionates to bromine gas and bromine tri- and pentafluorides. Use the following to find Δfor the decomposition of BrF to its elements:
Which member of each pair has more covalentcharacter in
its bonds: (a) LiCl or KCl; (b) AlCl3 or PCl3; (c) NCl3 or AsCl3?
Select the stronger bond in each pair:
(a) Cl-Cl or Br-Br
(b) Br-Br or I-I
(c) F-F or Cl-Cl.
Why doesn't the F-F bond strength follow the group trend?
What do you think about this solution?
We value your feedback to improve our textbook solutions.