Chapter 14: Q14.97 (page 623)
Question: Complete and balance the following:
Short Answer
Answer
(a)
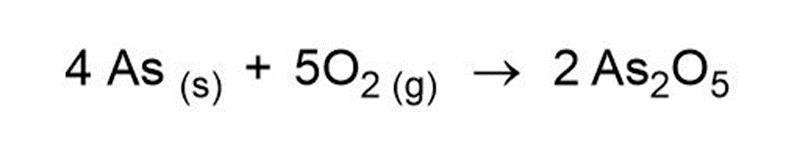
(b)
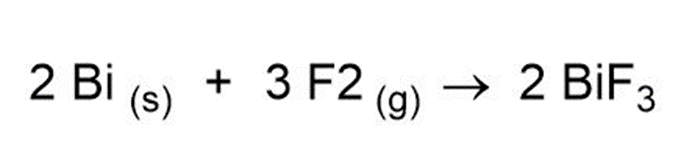
(c)
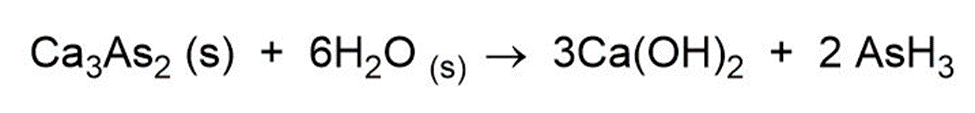
Learning Materials
Features
Discover
Chapter 14: Q14.97 (page 623)
Question: Complete and balance the following:
Answer
(a)
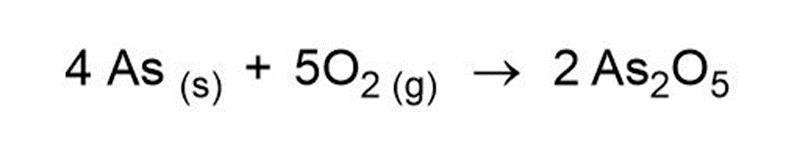
(b)
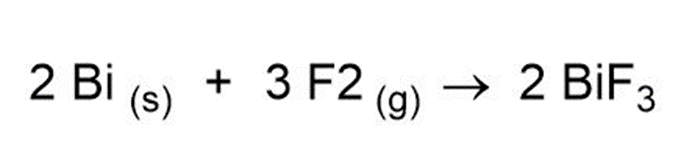
(c)
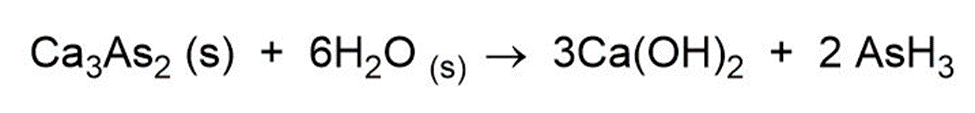
All the tools & learning materials you need for study success - in one app.
Get started for free
One similarity between B and Si is the explosive combustion of their hydrides in air. Write balanced equations for the combustion of androle="math" localid="1653907272447" .
Question: As you move down Group 5A (15), the melting points of the elements increases and then decreases. Explain
Boron nitride (BN) has a structure similar to graphite, but is a white insulator rather than a black conductor. It is synthesized by heating diboron trioxide with ammonia at about .
(a) Write a balanced equation for the formation of BN; water forms also.
(b) Calculate for the production of BN (of BN is ).
(c) Boron is obtained from the mineral borax, . How much borax is needed to produce 0.1kg of BN, assuming 72% yield?
The main reason alkali metal dihalides (MX2) do not form is the high IE2of the metal.
(a) Why is IE2 so high for alkali metals?
(b) The IE2 for Cs is 2255 kJ/mol, low enough for CsF2 to form exothermically . This compound cannot be synthesized, however, because CsF forms with a much greater release of heatlocalid="1663354802147" . Thus, the breakdown of CsF2 to CsF happens readily. Write the equation for this breakdown, and calculate the heat of reaction per mole of CsF.
Rank the following acids in order of decreasing acid strength: , , ,
What do you think about this solution?
We value your feedback to improve our textbook solutions.