Chapter 16: Q16.60P (page 731)
At , what is the fraction of collisions with energy equal to or greater than an activation energy of 100. kJ/mol?
Short Answer
The fraction of collisions is equal to
Learning Materials
Features
Discover
Chapter 16: Q16.60P (page 731)
At , what is the fraction of collisions with energy equal to or greater than an activation energy of 100. kJ/mol?
The fraction of collisions is equal to
All the tools & learning materials you need for study success - in one app.
Get started for free
Give two reasons to measure initialrates in a kinetic study.
For the reaction , [C] vs. time is plotted:
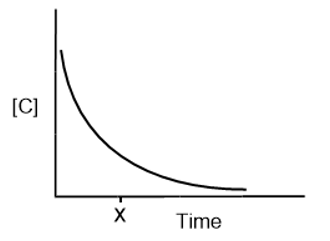
How do you determine each of the following?
(a) The average rate over the entire experiment
(b) The reaction rate at time x
(c) The initial reaction rate
(d) Would the values in parts (a), (b) and (c) be different if you plotted [D] vs. time? Explain.
Assume water boils at in Houston (near sea level), and at in Cripple Creek, Colorado (near 9500 ft). If it takes 4.8 min to cook an egg in Cripple Creek and 4.5 min in Houston, what is for this process?
Reaction rate is expressed in terms of changes in the concentration of reactants andproducts. Write a balanced equation for
Rate
The rate constant of a reaction is at , and the activation energy is 33.6 kJ/mol. What is k at ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.