Chapter 16: Q16.11 P (page 728)
For the reaction , [C] vs. time is plotted:
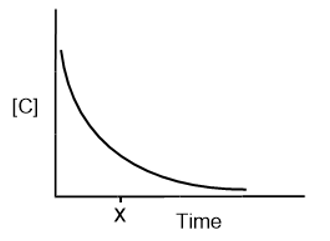
How do you determine each of the following?
(a) The average rate over the entire experiment
(b) The reaction rate at time x
(c) The initial reaction rate
(d) Would the values in parts (a), (b) and (c) be different if you plotted [D] vs. time? Explain.
Short Answer
The answer is,
(a) 1
(b)
(c) 0
(d) (a) and (c) are same, but (b) will belocalid="1663360591475"

