Chapter 16: Q16.10 P (page 728)
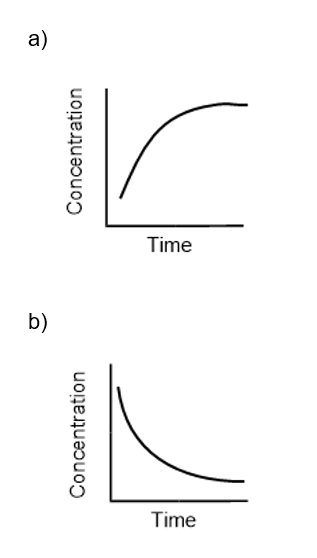
For the reaction , sketch two curves on the same set of axes that show (a) The formation of product as a function of time(b) The consumption of reactant as a function of time
Short Answer
The answer is,
Learning Materials
Features
Discover
Chapter 16: Q16.10 P (page 728)
For the reaction , sketch two curves on the same set of axes that show (a) The formation of product as a function of time(b) The consumption of reactant as a function of time
The answer is,

All the tools & learning materials you need for study success - in one app.
Get started for free
Give two reasons to measure initialrates in a kinetic study.
To determine its rate law. Assuming that you have a valid experimental procedure for obtaining [A2] and [B2] at various times, explain how you determine
(a) the initial rate,
(b) the reaction orders, and
(c) the rate constant.
The molecular scenes below represent the first-order reaction as cyclopropane (red) is converted to propene (green):
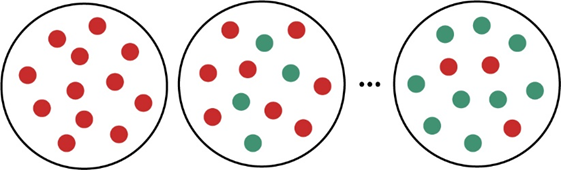
Determine (a) the half-life and (b) the first-order rate constant.
a) For a reaction with a given Ea, how does an increase in T affect the rate?
(b) For a reaction at a given T, how does a decrease in Ea affect the rate?
Define reaction rate, assuming constant temperature and a closed reaction vessel, why does the rate change with time?
What do you think about this solution?
We value your feedback to improve our textbook solutions.