Chapter 16: Q16.118 CP (page 735)
The molecular scenes below represent the first-order reaction as cyclopropane (red) is converted to propene (green):
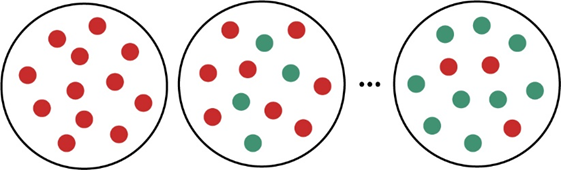
Determine (a) the half-life and (b) the first-order rate constant.
Short Answer
Half-life is
b. Rate constant for a reaction is

