Chapter 5: Q5.29P (page 228)
If of argon occupies a 75.0-mL container at , what is the pressure (in torr)?
Short Answer
Answer
The pressure of the gas is 365 torr.
Learning Materials
Features
Discover
Chapter 5: Q5.29P (page 228)
If of argon occupies a 75.0-mL container at , what is the pressure (in torr)?
Answer
The pressure of the gas is 365 torr.
All the tools & learning materials you need for study success - in one app.
Get started for free
Convert the following:
(a) 76.8 cmHg to atm (b) 27.5 atm to kPa
(c) 6.50 atm to bar (d) 0.937 kPa to torr
A sample of Freon-12 () occupies 25.5 L at 298 K and 153.3 kPa. Find its volume at STP.
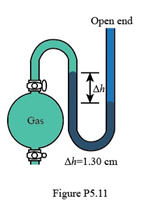
In Figure P5.11, what is the pressure of the gas in the flask (in kPa) if the barometer reads 765.2 mmHg?
In preparation for a demonstration, your professor brings a 1.5-L bottle of sulfur dioxide into the lecture hall before class to allow the gas to reach room temperature. If the pressure gauge reads 85 psi and the temperature in the hall is , how many moles of sulfur dioxide are in the bottle?
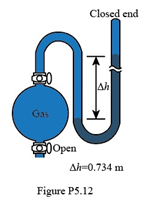
If the sample flask in Figure P5.12 is open to the air, what is the atmospheric pressure (in atm)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.