Chapter 5: Q5.28P (page 228)
A sample of chlorine gas is confined in a 5.0-L container at 328 torr and . How many moles of gas are in the sample?
Short Answer
Answer
The number of moles of gas in the sample is 0.085 mol.
Learning Materials
Features
Discover
Chapter 5: Q5.28P (page 228)
A sample of chlorine gas is confined in a 5.0-L container at 328 torr and . How many moles of gas are in the sample?
Answer
The number of moles of gas in the sample is 0.085 mol.
All the tools & learning materials you need for study success - in one app.
Get started for free
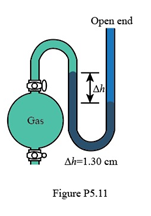
In Figure P5.11, what is the pressure of the gas in the flask (in kPa) if the barometer reads 765.2 mmHg?
In the following relationships, which quantities are variables and which are fixed: (a) Charles’s law; (b) Avogadro’s law; (c) Amontons’s law?
Convert the following:
(a) 0.745 atm to mmHg (b) 992 torr to bar
(c) 365 kPa to atm (d) 804 mmHg to kPa
To collect a beaker of gas by displacing the air already in the beaker, would you hold the beaker upright or inverted? Why? How would you hold the beaker to collect ?
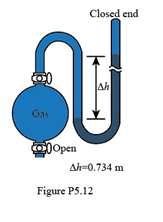
If the sample flask in Figure P5.12 is open to the air, what is the atmospheric pressure (in atm)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.