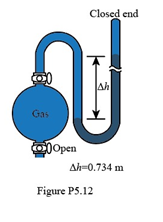
Chapter 5: Q5.12P (page 227)
If the sample flask in Figure P5.12 is open to the air, what is the atmospheric pressure (in atm)?
Short Answer
The atmospheric pressure is 0.966 atm.
Learning Materials
Features
Discover
Chapter 5: Q5.12P (page 227)
If the sample flask in Figure P5.12 is open to the air, what is the atmospheric pressure (in atm)?

The atmospheric pressure is 0.966 atm.
All the tools & learning materials you need for study success - in one app.
Get started for free
A sample of sulfur hexafluoride gas occupies 9.10 L at . Assuming that the pressure remains constant, what temperature (in ) is needed to reduce the volume to 2.50 L?
A sample of carbon monoxide occupies 3.65 L at 298 K and 745 torr. Find its volume at and 367 torr.
Are the particles in a gas farther apart or closer together than the particles in a liquid? Use your answer to explain each of the following general observations:
(a) Gases are more compressible than liquids.
(b) Gases have lower viscosities than liquids.
(c) After thorough stirring, all gas mixtures are solutions.
(d) The density of a substance in the gas state is lower than in the liquid state.
Convert the following:
(a) 76.8 cmHg to atm (b) 27.5 atm to kPa
(c) 6.50 atm to bar (d) 0.937 kPa to torr
A 93-L sample of dry air cools from to while the pressure is maintained at 2.85 atm. What is the final volume?
What do you think about this solution?
We value your feedback to improve our textbook solutions.