Chapter 5: Q5.13P (page 227)
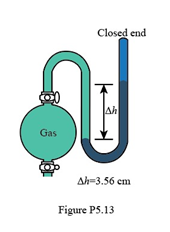
What is the pressure (in Pa) of the gas in the flask in Figure P5.13?
Short Answer
The pressure of the gas is .
Learning Materials
Features
Discover
Chapter 5: Q5.13P (page 227)
What is the pressure (in Pa) of the gas in the flask in Figure P5.13?

The pressure of the gas is .
All the tools & learning materials you need for study success - in one app.
Get started for free
You have 357 mL of chlorine trifluoride gas at 699 mmHg and . What is the mass (in g) of the sample?
How does a sample of gas differ in its behavior from a sample of liquid in each of the following situations?
(a) The sample is transferred from one container to a larger one.
(b) The sample is heated in an expandable container, but no change of state occurs.
(c) The sample is placed in a cylinder with a piston, and an external force is applied.
What is the effect of the following on the volume of 1 mol of an ideal gas?
(a) Temperature decreases from 800 K to 400 K (at constantP).
(b) Temperature increases from to (at constantP).
(c) Pressure increases from 2 atm to 6 atm (at constant T).
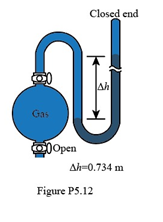
If the sample flask in Figure P5.12 is open to the air, what is the atmospheric pressure (in atm)?
On a cool, rainy day, the barometric pressure is 730 mmHg. Calculate the barometric pressure in centimeters of water (cmH2O) (dof Hg = 13.5 g/mL; dof H2O = 1.00 g/mL).
What do you think about this solution?
We value your feedback to improve our textbook solutions.