Chapter 5: Q5.26P (page 228)
A sample of Freon-12 () occupies 25.5 L at 298 K and 153.3 kPa. Find its volume at STP.
Short Answer
Answer
The volume at STP is 35.4 L.
Learning Materials
Features
Discover
Chapter 5: Q5.26P (page 228)
A sample of Freon-12 () occupies 25.5 L at 298 K and 153.3 kPa. Find its volume at STP.
Answer
The volume at STP is 35.4 L.
All the tools & learning materials you need for study success - in one app.
Get started for free
A sample of carbon monoxide occupies 3.65 L at 298 K and 745 torr. Find its volume at and 367 torr.
How does a sample of gas differ in its behavior from a sample of liquid in each of the following situations?
(a) The sample is transferred from one container to a larger one.
(b) The sample is heated in an expandable container, but no change of state occurs.
(c) The sample is placed in a cylinder with a piston, and an external force is applied.
In the following relationships, which quantities are variables and which are fixed: (a) Charles’s law; (b) Avogadro’s law; (c) Amontons’s law?
What is the effect of the following on the volume of 1 mol of an ideal gas?
(a) Half the gas escapes (at constantPandT).
(b) The initial pressure is 722 torr, and the final pressure is 0.950 atm; the initial temperature is 32oF, and the final temperature is 273 K.
(c) Both the pressure and temperature decrease to one-fourth of their initial values.
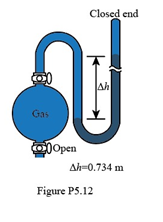
If the sample flask in Figure P5.12 is open to the air, what is the atmospheric pressure (in atm)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.