Chapter 17: Q17.92CP (page 779)
Use each of the following reaction quotients to write the balanced equation:
(a)
(b)
Short Answer
a) The balanced equation is .
b) The balanced equation is .
Learning Materials
Features
Discover
Chapter 17: Q17.92CP (page 779)
Use each of the following reaction quotients to write the balanced equation:
(a)
(b)
a) The balanced equation is .
b) The balanced equation is .
All the tools & learning materials you need for study success - in one app.
Get started for free
An industrial chemist introduces ofandofinto acontainer atand then raises the temperature to, at which:
How many grams ofare present at equilibrium?
The "filmstrip" represents five molecular scenes of a gaseous mixture as it reaches equilibrium over time:
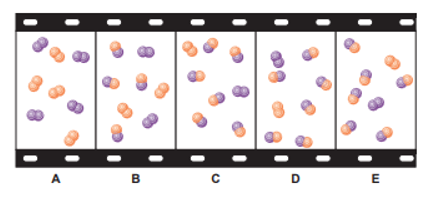
X is purple and Y is orange: .
(a) Write the reaction quotient, Q, for this reaction.
(b) If each particle represents, find Q for each scene.
(c) If K>1, is time progressing to the right or to the left? Explain.
(d) Calculate K at this temperature.
(e) If, which scene, if any, best represents the mixture at a higher temperature? Explain.
(f) Which scene, if any, best represents the mixture at a higher pressure (lower volume)? Explain.
The oxidation of is the key step in production:role="math" localid="1654927501262" (a) What qualitative combination of T and P maximizes yield? (b) How does addition of affect Q? K? (c) Why is catalysis used for this reaction?
Balance each reaction and write its reaction quotient, :
Does Q for the formation of 1 mol of NO from its elements differ from Q for the decomposition of 1 mol of NO to its elements? Explain and give the relationship between the two Q’s.
What do you think about this solution?
We value your feedback to improve our textbook solutions.