Chapter 17: 17.86_CP (page 779)
An industrial chemist introduces ofandofinto acontainer atand then raises the temperature to, at which:
How many grams ofare present at equilibrium?
Short Answer
The amount of are present at equilibrium is .
Learning Materials
Features
Discover
Chapter 17: 17.86_CP (page 779)
An industrial chemist introduces ofandofinto acontainer atand then raises the temperature to, at which:
How many grams ofare present at equilibrium?
The amount of are present at equilibrium is .
All the tools & learning materials you need for study success - in one app.
Get started for free
The "filmstrip" represents five molecular scenes of a gaseous mixture as it reaches equilibrium over time:
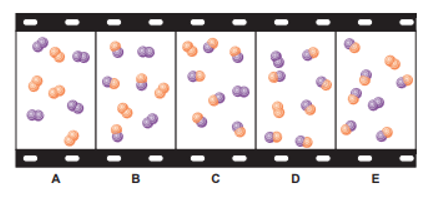
X is purple and Y is orange: .
(a) Write the reaction quotient, Q, for this reaction.
(b) If each particle represents, find Q for each scene.
(c) If K>1, is time progressing to the right or to the left? Explain.
(d) Calculate K at this temperature.
(e) If, which scene, if any, best represents the mixture at a higher temperature? Explain.
(f) Which scene, if any, best represents the mixture at a higher pressure (lower volume)? Explain.
When of and mol of are placed in an evacuated sealed container and heated to , after equilibrium is established:
An additional of is pumped in. What is the total mass (inrole="math" localid="1656942360456" ) of after equilibrium is re-established?
For a problem involving the catalysed reaction of methane and
steam, the following reaction table was prepared:
Explain the entries in the “Change” and “Equilibrium” rows.
Does Q for the formation of 1 mol of from differs from Q for the formation of from and 1 mol of ? Explain and give the relationship between the two Q’s
A key step in the extraction of iron from its or
This step occurs in the C to zone within a blast furnace. What are the equilibrium partial pressures of CO(g) and (g) when 1.00 atm of CO(g) and excess FeO(s) react in a sealed container at ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.