Chapter 17: Q13P (page 775)
Balance each reaction and write its reaction quotient, :
Short Answer
The answer is,
Learning Materials
Features
Discover
Chapter 17: Q13P (page 775)
Balance each reaction and write its reaction quotient, :
The answer is,
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Calculate Kc for each of the following equilibria:
At for the reaction
role="math" localid="1655096121156"
In one experiment, 0.20 atm of HBr(g), 0.010 atm of , and 0.010 atm of are introduced into a container. Is the reaction at equilibrium? If not, in which direction will it proceed?
The "filmstrip" represents five molecular scenes of a gaseous mixture as it reaches equilibrium over time:
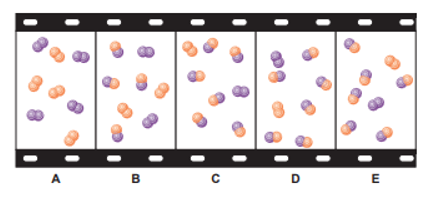
X is purple and Y is orange: .
(a) Write the reaction quotient, Q, for this reaction.
(b) If each particle represents, find Q for each scene.
(c) If K>1, is time progressing to the right or to the left? Explain.
(d) Calculate K at this temperature.
(e) If, which scene, if any, best represents the mixture at a higher temperature? Explain.
(f) Which scene, if any, best represents the mixture at a higher pressure (lower volume)? Explain.
Does Q for the formation of 1 mol of from differs from Q for the formation of from and 1 mol of ? Explain and give the relationship between the two Q’s
Highly toxic disulfurdecafluoride decomposes by a free radical process: . In a study of the decomposition, was placed in a - flask and heated to was at equilibrium. More was added, and when equilibrium was retained, was . How diddata-custom-editor="chemistry" and change from the original to the new equilibrium position after the addition of more data-custom-editor="chemistry" ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.