Chapter 17: Q12CP (page 775)
Balance each reaction and write its reaction quotient, :
Short Answer
The answer is,
Learning Materials
Features
Discover
Chapter 17: Q12CP (page 775)
Balance each reaction and write its reaction quotient, :
The answer is,
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider this equilibrium system:
How does the equilibrium position shift as a result of each of the following disturbances? (a) is added.
(b) is removed by adding solid .
(c) Additional is added to the system.
(d) Dry ice is added at constant temperature.
What does “disturbance” mean in Le Châtelier’s principle?
Scenes A, B, and C below depict this reaction at three temperatures:
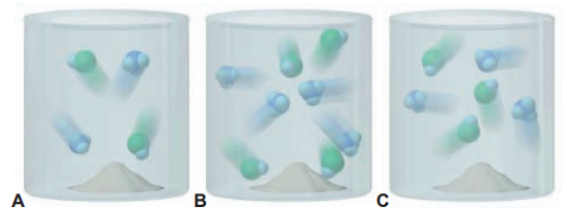
(a) Which best represents the reaction mixture at the highest temperature? Explain. (b) Which best represents the reaction mixture at the lowest temperature? Explain.
In the 1980s, CFC-11 was one of the most heavily produced chlorofluorocarbons. The last step in its formation is
If you start the reaction with equal concentrations of and HF, you obtain
equal concentrations of and HCl at equilibrium. Are the final
concentrations of and HCl equal if you start with unequal
concentrations of and HF? Explain.
When of and mol of are placed in an evacuated sealed container and heated to , after equilibrium is established:
An additional of is pumped in. What is the total mass (inrole="math" localid="1656942360456" ) of after equilibrium is re-established?
What do you think about this solution?
We value your feedback to improve our textbook solutions.