Chapter 18: Q18.90P (page 826)
What is the pH of 0.070 M dimethylamine?
Short Answer
Answer
The value of pHis obtained as: 11.81
Learning Materials
Features
Discover
Chapter 18: Q18.90P (page 826)
What is the pH of 0.070 M dimethylamine?
Answer
The value of pHis obtained as: 11.81
All the tools & learning materials you need for study success - in one app.
Get started for free
Human urine has a normal pH of 6.2. If a person eliminates an average of 1250mL of urine per day, how manyions are eliminated per week?
Question: Which of the following are Arrhenius bases?
(a)
(b)
(c)
(d)
Question: Write the expression for each of the following in water:
(a)
(b)
(c)
Hemoglobin (Hb) transports oxygen in the blood:
In blood,is held nearly constant at.
(a) How does the equilibrium position change in the lungs?
(b) How does it change in O2-deficient cells?
(c) Excessive vomiting may lead to metabolic alkalosis, in which [] in blood decreases. How does this condition affect the ability of Hb to transport?
(d) Diabetes mellitus may lead to metabolic acidosis, in which [] in blood increases. How does this condition affect the ability of Hb to transport?
The antimalarial properties of quinine () saved thousands of lives during construction of the Panama Canal. This substance is a classic example of the medicinal wealth of tropical forests. Both N atoms are basic, but the N (coloured) of the 3amine group is far more basic (pKb =5.1) than the N within the aromatic ring system (pKb =9.7).
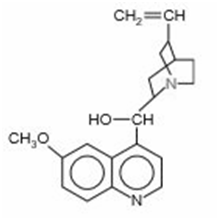
(a) A saturated solution of quinine in water is only. What is the pH of this solution?
(b) Show that the aromatic N contributes negligibly to the pH of the solution.
(c) Because of its low solubility, quinine is given as the salt quinine hydrochloride,which is 120times more soluble than quinine. What is the pH ofM quinine hydrochloride?
(d) An antimalarial concentration in water is 15% quinine hydrochloride by mass (d 1.0g/mL). What is the pH?
What do you think about this solution?
We value your feedback to improve our textbook solutions.