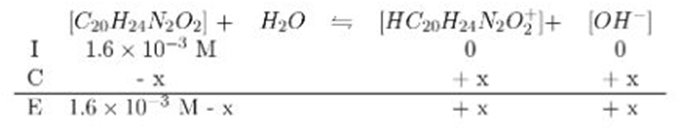Chapter 18: Q18.187CP (page 830)
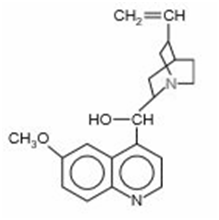
The antimalarial properties of quinine () saved thousands of lives during construction of the Panama Canal. This substance is a classic example of the medicinal wealth of tropical forests. Both N atoms are basic, but the N (coloured) of the 3amine group is far more basic (pKb =5.1) than the N within the aromatic ring system (pKb =9.7).
(a) A saturated solution of quinine in water is only. What is the pH of this solution?
(b) Show that the aromatic N contributes negligibly to the pH of the solution.
(c) Because of its low solubility, quinine is given as the salt quinine hydrochloride,which is 120times more soluble than quinine. What is the pH ofM quinine hydrochloride?
(d) An antimalarial concentration in water is 15% quinine hydrochloride by mass (d 1.0g/mL). What is the pH?
Short Answer
- The ions produced by the aromatic amine do not affect the overall total of ions significantly
- The pH of quinine hydrochloride is role="math" localid="1663266152189" .