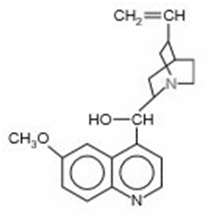
Chapter 18: Q18.179CP (page 829)
In his acid-base studies, Arrhenius discovered an important fact involving reactions like the following:
(a) Complete the reactions and use the data for the individual ions in Appendix B to calculate each
(b) Explain your results and use them to predict for
Short Answer
a) For the reaction of NaOH and HCI,
b)