Chapter 18: Q18.9P (page 823)
Write the expression for each of the following in water:
(a)
(b)
(c)
Short Answer
(a) The expression for is .
(b) The expression for is .
(c) The expression for is .
Learning Materials
Features
Discover
Chapter 18: Q18.9P (page 823)
Write the expression for each of the following in water:
(a)
(b)
(c)
(a) The expression for is .
(b) The expression for is .
(c) The expression for is .
All the tools & learning materials you need for study success - in one app.
Get started for free
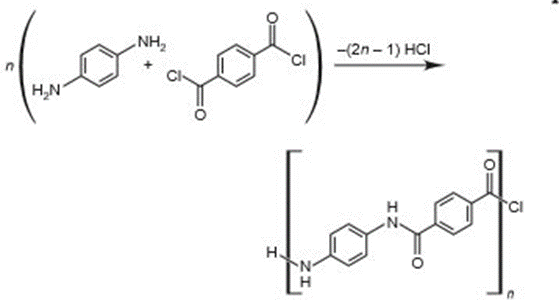
Kevlar®, a polymeric aromatic amide, is synthesized from the monomers p-phenylenediamine (left) and terephthaloyl chloride (right). The polymer strands are initially aligned randomly, but they are dissolved and spun to give a lightweight, flexible product that is five times stronger than steel of the same weight
The first step in the reaction of an amine with an acid chloride is
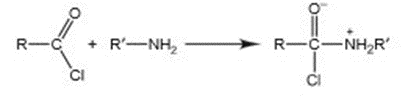
(a) Describe this step in terms of a Lewis acid-base reaction.
(b) Would you expect water or ammonia to react faster with an acid chloride? Explain
(a) What is the pH of M HI? Is the solution neutral, acidic, or basic?
(b) What is the pOH of 2.55 M Ba ? Is the solution neutral, acidic, or basic?
Write the expression for each of the following in water:
(a)
(b)
(c)
Auto ionization occurs in methanol and in ethylenediamine ().
(a) The auto ionization constant of methanolis . What is in pure ?
(b) The concentration ofin pure is. What is the auto ionization constant of ethylenediamine?
Drinking water is often disinfected with , which hydrolyzes to form HClO, a weak acid but powerful disinfectant:
The fraction of HClO in solution is defined as
(a) What is the fraction of HClO at pH 7.00(Ka of HClO=?
(b) What is the fraction at pH 10.00?
What do you think about this solution?
We value your feedback to improve our textbook solutions.