Chapter 9: Q. 9.104 (page 318)
How many liters of a solution will provide of
Short Answer
of a solution provideof.
Learning Materials
Features
Discover
Chapter 9: Q. 9.104 (page 318)
How many liters of a solution will provide of
of a solution provideof.
All the tools & learning materials you need for study success - in one app.
Get started for free
Indicate whether aqueous solutions of each of the following solutes contain only ions, only molecules, or mostly molecules and a few ions:
a. Acetic acid, , a weak electrolyte.
b. , a strong electrolyte.
c. fructose,, a nonelectrolyte.
Select the diagram that represents the shape of a red blood cell when placed in each of the following a to


a. solution
b. localid="1654008537048" glucose solution
c. localid="1654008233523" solution
d. localid="1654008237506" glucose solution
e. localid="1654008241312" glucose solution
An intravenous solution of mannitol is used as a diuretic to increase the loss of sodium and chloride by a patient. If a patient receives of a mannitol solution, how many grams of mannitol were given?
Classify the solute represented in each of the following equations as a strong, weak, or nonelectrolyte:
a.
b.
c.
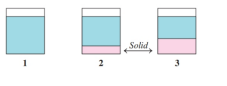
If all the solute is dissolved in diagram , how would heating or cooling the solution cause each of the following changes? (9.3)
a.
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.