Chapter 2: Q3P (page 40)
Question: Approximately how many molecules of H2Oare in one spoonful of water, assuming that the spoon holds about 18 mL?
Short Answer
Answer
The molecules H2O that are in one spoonful of water is .
Learning Materials
Features
Discover
Chapter 2: Q3P (page 40)
Question: Approximately how many molecules of H2Oare in one spoonful of water, assuming that the spoon holds about 18 mL?
Answer
The molecules H2O that are in one spoonful of water is .
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the relationship between the strength of an acid and its pK value?
Marble is composed largely of calcium carbonate (CaCO3). Spilling tomato juice on a slab of marble etches (eats away) the surface. Explain what happens in chemical terms.
Question: Explain why water forms nearly spherical droplets on the surface of a freshly waxed car. Why doesn’t water bead on a clean windshield?
Question: Draw the structures of the conjugate bases of the following acids:
a.
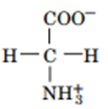
b.
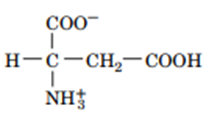
Patients with kidney failure frequently develop metabolic acidosis. If such patients undergo dialysis, the dialysate includes sodium bicarbonate at a concentration higher than that of the blood. Explain why this would benefit the patient.
What do you think about this solution?
We value your feedback to improve our textbook solutions.