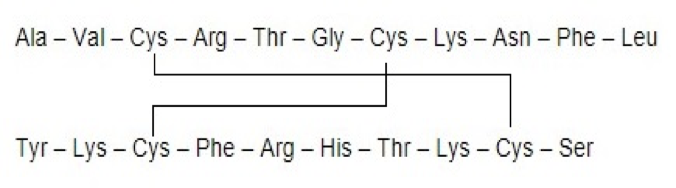
Chapter 5: Q33P. (page 129)
Treatment of a polypeptide with 2-mercaptoethanol yields two polypeptides:
1. Ala–Val–Cys–Arg–Thr–Gly–Cys–Lys–Asn–Phe–Leu
2. Tyr–Lys–Cys–Phe–Arg–His–Thr–Lys–Cys–Ser
Treatment of the intact polypeptide with trypsin yields fragments with the following amino acid compositions:
3. (Ala, Arg, Cys2, Ser, Val)
4. (Arg, Cys2, Gly, Lys, Thr, Phe)
5. (Asn, Leu, Phe)
6. (His, Lys, Thr)
7. (Lys, Tyr)
Indicate the positions of the disulfide bonds in the intact polypeptide.
Short Answer
The disulfide bond formed between the two peptides is shown below.