Chapter 14: Q5P (page 475)
Rank the following compounds in order of increasing oxidation state.
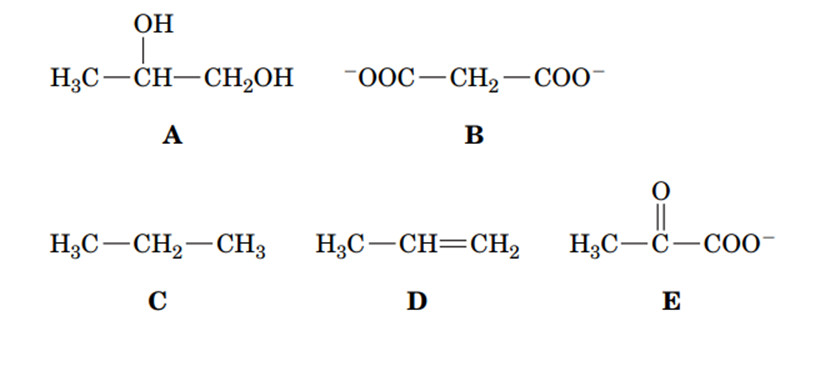
Short Answer
The ranking in increasing oxidation state is C D A E B.
Learning Materials
Features
Discover
Chapter 14: Q5P (page 475)
Rank the following compounds in order of increasing oxidation state.

The ranking in increasing oxidation state is C D A E B.
All the tools & learning materials you need for study success - in one app.
Get started for free
Assuming 100% efficiency of energy conservation, how many moles of ATP can be synthesized under standard conditions by the complete oxidation of 1 mol of glucose?
The reaction catalyzed by malate dehydrogenase,
has a value of . (a) Would this reaction occur spontaneously in a cell? (b) How does the citrate synthase reaction (described in Problem 7 ) promote the malate dehydrogenase reaction in the cell? What is the overall change in free energy for the two reactions?
Question: If intracellular , and , calculate the concentration of AMP at pH 7 and under the condition that the adenylate kinase reaction is at equilibrium.
How is Δℰrelated to ΔG?
Explain why cadmium and mercury are toxic to most organisms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.