Chapter 14: Q6P (page 476)
In the partial reactions shown below, is the reactant undergoing oxidation or reduction?
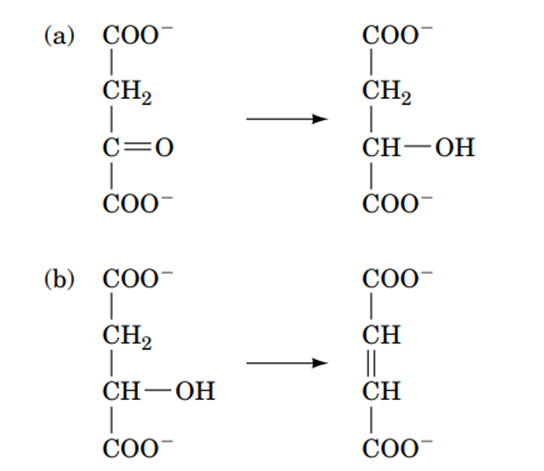
Short Answer
(a) The reactant is undergoing reduction.
(b) The reactant is undergoing reduction.
Learning Materials
Features
Discover
Chapter 14: Q6P (page 476)
In the partial reactions shown below, is the reactant undergoing oxidation or reduction?

(a) The reactant is undergoing reduction.
(b) The reactant is undergoing reduction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Citrate synthase catalyses the reaction
The standard free energy change for the reaction is role="math" localid="1660738017563" . (a) Calculate the equilibrium constant for this reaction at 37oC. (b) Would you expect this reaction to serve as a control point for its pathway (the citric acid cycle)?
Question: Predict whether creatine kinase will operate in the direction of ATP synthesis or phosphocreatine synthesis at when [ATP] , [ADP] , [phosphocreatine] , and [creatine] .
The reaction catalyzed by malate dehydrogenase,
has a value of . (a) Would this reaction occur spontaneously in a cell? (b) How does the citrate synthase reaction (described in Problem 7 ) promote the malate dehydrogenase reaction in the cell? What is the overall change in free energy for the two reactions?
Rank the following compounds in order of increasing oxidation state.
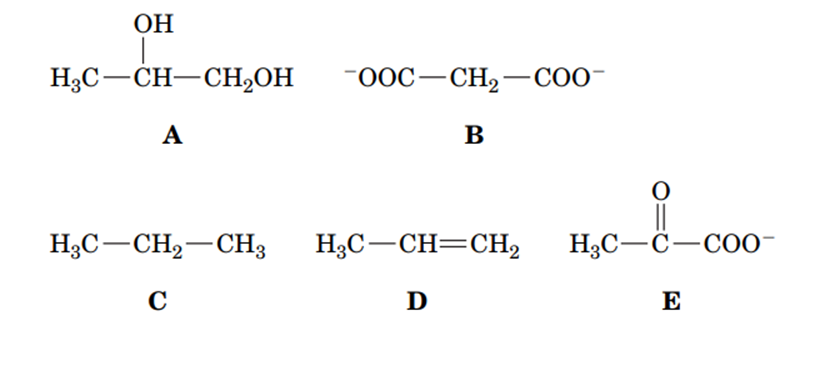
Why might transcriptomic and proteomic analyses reveal different information about the metabolic activity of a particular tissue?
What do you think about this solution?
We value your feedback to improve our textbook solutions.