Chapter 21: Q9. (page 771)
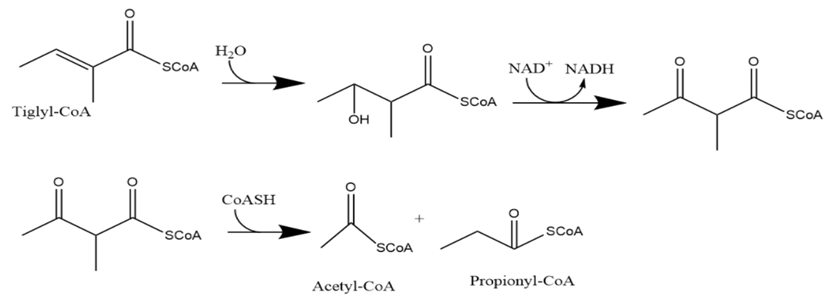
In the degradation pathway for isoleucine (Fig. 21-21), draw the reactions that convert tigyl CoAto acetyl CoA andpropionyl CoA.
Short Answer
Learning Materials
Features
Discover
Chapter 21: Q9. (page 771)
In the degradation pathway for isoleucine (Fig. 21-21), draw the reactions that convert tigyl CoAto acetyl CoA andpropionyl CoA.

All the tools & learning materials you need for study success - in one app.
Get started for free
How do anammox bacteria participate in the nitrogen cycle? Where do the organisms live and how do they contain the toxic intermediates of their specialized metabolism?
An L-amino acid oxidase in mammalian peroxisomes requires and as substrates and catalyzes amino acid deamination, producing as a product. Write a balanced equation for the reaction.
What are the substrates and products of the nitric oxide synthase reaction?
(a) How many ATP equivalents are consumed by the reactions of the urea cycle? (b) Operation of the urea cycle actually generates more ATP than it consumes. Explain.
Describe howα-ketoglutarate and oxaloacetate participate in amino acid catabolism.
What do you think about this solution?
We value your feedback to improve our textbook solutions.