Chapter 15: Q18P (page 412)
Question:(I) A heat engine exhausts 8200 J of heat while performing 2600 J of useful work. What is the efficiency of this engine?
Short Answer
The efficiency of the heat engine is \(24.07\% \).
Learning Materials
Features
Discover
Chapter 15: Q18P (page 412)
Question:(I) A heat engine exhausts 8200 J of heat while performing 2600 J of useful work. What is the efficiency of this engine?
The efficiency of the heat engine is \(24.07\% \).
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:(I) The exhaust temperature of a heat engine is 230°C. What is the high temperature if the Carnot efficiency is 34%?
(I) One liter of air is cooled at constant pressure until its volume is halved, and then it is allowed to expand isothermally back to its original volume. Draw the process on a PV diagram.
Question: An ideal monatomic gas is contained in a tall cylindrical jar of cross-sectional area \({\bf{0}}{\bf{.080}}\;{{\bf{m}}^{\bf{2}}}\) fitted with an airtight frictionless 0.15-kg movable piston. When the gas is heated (at constant pressure) from 25°C to 55°C, the piston rises 1.0 cm. How much heat was required for this process? Assume atmospheric pressure outside. (Hint: See Section 14–2.)
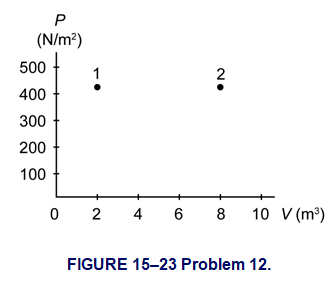
Question: (III) The PV diagram in Fig. 15–23 shows two possible states of a system containing 1.75 moles of a monatomic ideal gas. \(\left( {{P_1} = {P_2} = {\bf{425}}\;{{\bf{N}} \mathord{\left/{\vphantom {{\bf{N}} {{{\bf{m}}^{\bf{2}}}}}} \right.} {{{\bf{m}}^{\bf{2}}}}},\;{V_1} = {\bf{2}}{\bf{.00}}\;{{\bf{m}}^{\bf{3}}},\;{V_2} = {\bf{8}}{\bf{.00}}\;{{\bf{m}}^{\bf{3}}}.} \right)\) (a) Draw the process which depicts an isobaric expansion from state 1 to state 2, and label this process A. (b) Find the work done by the gas and the change in internal energy of the gas in process A. (c) Draw the two-step process which depicts an isothermal expansion from state 1 to the volume \({V_2}\), followed by an isovolumetric increase in temperature to state 2, and label this process B. (d) Find the change in internal energy of the gas for the two-step process B.
Question: (II) How much less per year would it cost a family to operate a heat pump that has a coefficient of performance of 2.9 than an electric heater that costs \(2000 to heat their home for a year? If the conversion to the heat pump costs \)15,000, how long would it take the family to break even on heating costs? How much would the family save in 20 years?
What do you think about this solution?
We value your feedback to improve our textbook solutions.