Chapter 4: Q58E (page 138)
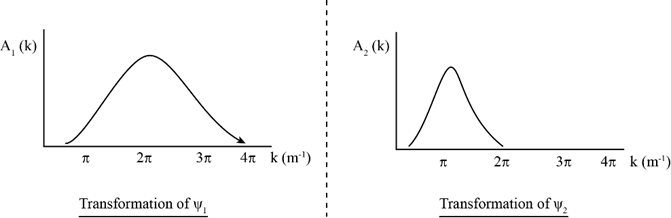
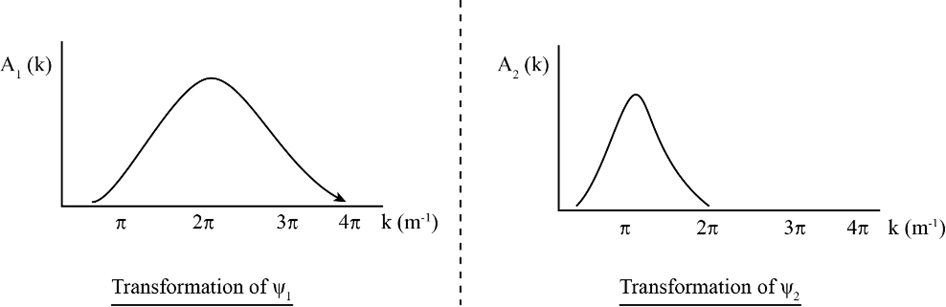
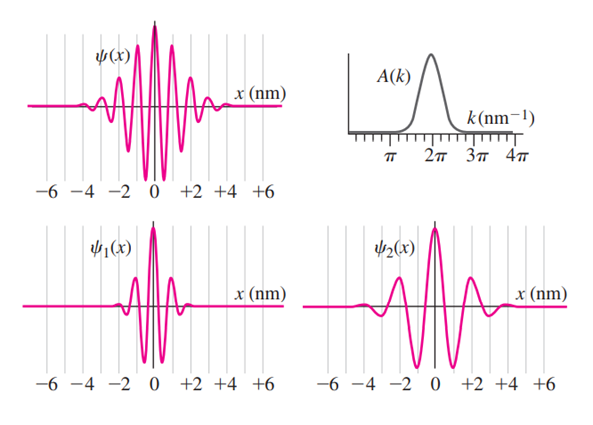
The top two plots in the accompanying diagram show a Gaussian wave functionand its Fourier transform. (a) How does theplot demonstrate that the particle’s momentum is roughly? (b) How does theplot demonstrate the same thing? (c) If you measured the particle’s momentum,would be a likely value. Wouldbe a likely value? Would? Would? (d) The bottom plots show two different Gaussian wave functions. Make sketches of their corresponding Fourier transforms.
Short Answer
(a) Since, the wavelength is 1 nm, so, the particle’s momentum is.
(b) The graph is peaked at, so, the particle’s momentum is.
(c) The valuesandwould be likely values and would be unlikely value.
(d) The Fourier transforms sketches are as follows.