Chapter 4: Q39E (page 136)
Verify the claim made in a section 4.4 that if all results of a repeated experiment are equal, the standard deviation, equation (4.13) Will be0.
Short Answer
The standard deviation is 0.
Learning Materials
Features
Discover
Chapter 4: Q39E (page 136)
Verify the claim made in a section 4.4 that if all results of a repeated experiment are equal, the standard deviation, equation (4.13) Will be0.
The standard deviation is 0.
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the Compton wavelength of the electron, defined to be the wavelength it would have if its momentum were.
A beam of electrons strikes a barrier with two narrow but equal-width slits. A screen is located beyond the barrier. And electrons are detected as they strike the screen. The "center" of the screen is the point equidistant from the slits. When either slit alone is open,electrons arrive per second in a very small region at the center of the screen. When both slits are open, how many electrons will arrive per second in the same region at the center of the screen?
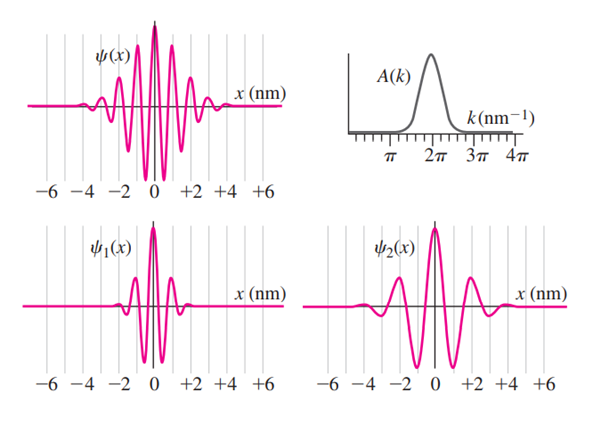
The top two plots in the accompanying diagram show a Gaussian wave functionand its Fourier transform. (a) How does theplot demonstrate that the particle’s momentum is roughly? (b) How does theplot demonstrate the same thing? (c) If you measured the particle’s momentum,would be a likely value. Wouldbe a likely value? Would? Would? (d) The bottom plots show two different Gaussian wave functions. Make sketches of their corresponding Fourier transforms.
(a) Experiment X is carried out nine times identically, and the value 5is obtained all nine times. Calculate the mean by definition (4-12). Then the standard deviation by definition (4-13). (b) Experiment Yis carried out nine times identically, and the integers 1 through 9 are each obtained once. Repeat the calculations of part (a) for this experiment. (c) For nine repetitions of the experimentZ.The tally is that 1, 5, and 9 are each obtained three times. Repeat the calculations. (d) Explain any differences between the results in parts (b) and (c). Is standard deviation a reasonable measure of spread?
According to the energy-time uncertainty principle, the lifetimeof a state and the uncertaintyin its energy are invertible proportional. Hydrogen's red spectral line is the result of an electron making a transition "downward" frum a quantum state whose lifetime is about.
(a) What inherent uncertainty in the energy of the emitted person does this imply? (Note: Unfortunately. we might use the symbol for the energy difference-i.e., the energy of the photon-but here it means the uncertainly in that energy difference.)
(b) To what range in wavelength s does this correspond? (As noted in Exercise 2.57. the uncertainty principle is one contributor to the broadening of spectral lines.)
(c) Obtain a general formula relating .
What do you think about this solution?
We value your feedback to improve our textbook solutions.