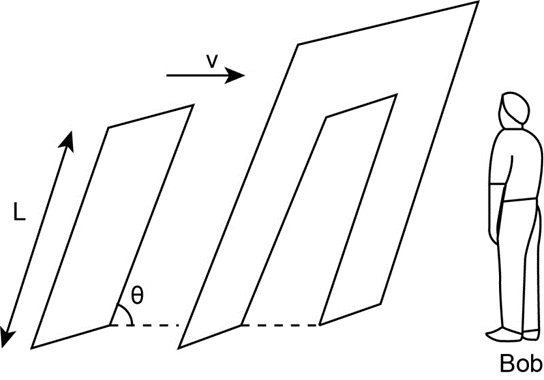
Bob, in frame , is observing the moving plank of Exercise 38. He quickly fabricates a wall, fixed in his frame, that has a hole of length L and that is slanted at angle, such that the plank will completely fill the hole as it passes through. This occurs at the instant t=0. According to Anna, moving with the plank, the plank is of course not of length L, but of length. Moreover, because Bob’s wall moves relative to her. Anna sees a hole that is less than L in length, a plank longer than L is headed toward a hole shorter than . Can the plank pass through the hole according to Anna? If so, at what time(s)? Explain.