Chapter 1: Q47E (page 1)
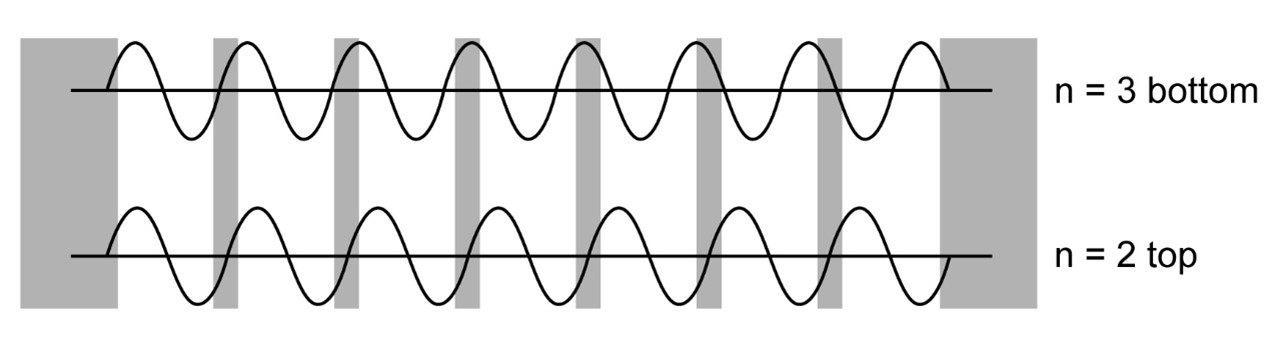
Make a rough sketches of the wave functions at the top of the band and the bottom of the band for a one dimensional “crystal” consisting of seven finite wells. Explain why these two states of roughly equal wave number have vastly different energies.
Short Answer
The sketch of the wavefunction is obtained and the explanation for the energy differences are explained.