Chapter 1: Q48E (page 1)
Question: Show that the normalization constant given in Table 7.3 for the angular parts of the wave function is correct.
Short Answer
Answer
It has been proved that the normalization for the case is correct.
Learning Materials
Features
Discover
Chapter 1: Q48E (page 1)
Question: Show that the normalization constant given in Table 7.3 for the angular parts of the wave function is correct.
Answer
It has been proved that the normalization for the case is correct.
All the tools & learning materials you need for study success - in one app.
Get started for free
Obtain a semi empirical binding energy per nucleon formula. Using this as a guide, explain why the Coulomb force, which is only aboutas strong as the inter nucleon attraction for two protons "in contact" (cf. Table 11.2), would eventually have to become a dominant factor in large nuclei. Assume that Z,N and A increase in rough proportion to one another.
A particle of mass m and energy E moving in a region where there is initially no potential energy encounters a potential dip of width L and depth .
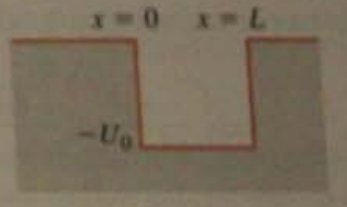
Show that the reflection probability is given by
(Hint: All that is needed is an appropriate substitution in a known probability.)
The allowed electron energies predicted by the Bohr model of the hydrogen atom are correct.(a) Determine the three lowest. (b) The electron can "jump" from a higher to lower energy. with a photon carrying away the energy difference. From the three energies found in part (a), determine three possible wavelengths of light emitted by a hydrogen atom.
Nuclei of the same mass number but different Zare known as isobars. Oxygen-15 and nitrogen- 15 are isobars.
(a) In which of the factors considered in nuclear binding (represented by terms in the semi empirical binding energy formula) do these two isobars differ?
(b) Which of the isobars should be more tightly bound?
(c) k your conclusion in part (b) supported by the decay mode information of Appendix 1? Explain.
(d) Calculate the binding energies of oxygen-15 and nitrogen-15. By how much do they differ?
(e) Repeat part (d) but use the semi empirical binding energy formula rather than the known atomic masses.
A function is nonzero only in the region of width centered at
where C is a constant.
(a) Find and plot versus the Fourier transform of this function.
(b) The function ) might represent a pulse occupying either finite distance (localid="1659781367200" position) or finite time (time). Comment on the wave number if is position and on the frequency spectrum if is time. Specifically address the dependence of the width of the spectrum on .
What do you think about this solution?
We value your feedback to improve our textbook solutions.