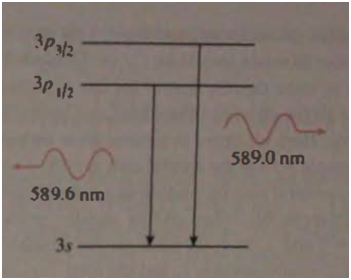
Chapter 1: Q32E (page 1)
Obtain a semi empirical binding energy per nucleon formula. Using this as a guide, explain why the Coulomb force, which is only aboutas strong as the inter nucleon attraction for two protons "in contact" (cf. Table 11.2), would eventually have to become a dominant factor in large nuclei. Assume that Z,N and A increase in rough proportion to one another.
Short Answer
The Coulomb term will dominate eventually.