Chapter 1: Q55E (page 1)
Verify that equation (4-19) follows from (4-16) and (4-18).
Short Answer
The proof of the equation is stated below.
Learning Materials
Features
Discover
Chapter 1: Q55E (page 1)
Verify that equation (4-19) follows from (4-16) and (4-18).
The proof of the equation is stated below.
All the tools & learning materials you need for study success - in one app.
Get started for free
Prove that fur any sine function of wavelength shorter than 2a, where ais the atomic spacing. there is a sine function with a wavelength longer than 2a that has the same values at the points x = a , 2a , 3a . and so on. (Note: It is probably easier to work with wave number than with wavelength. We sick to show that for every wave number greater than there is an equivalent less than .)
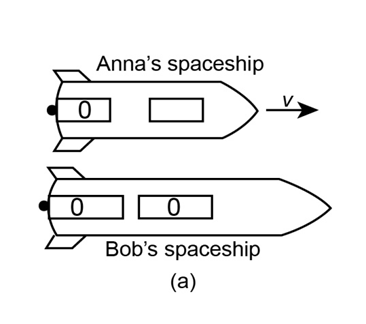
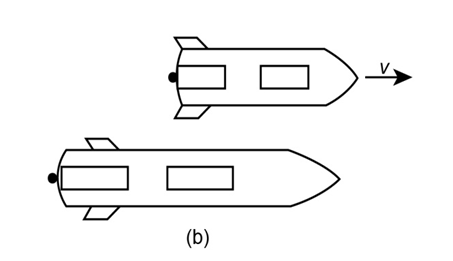
Anna and Bob have identical spaceship 60m long. The diagram shows Bob’s observations of Anna’s ship, which passes at a speed of . Clocks at the back of both ships read just as they pass. Bob is at the center of his ship and at t = 0 on his wrist watch peers at a second clock on Anna’s ship.
(a) What does this clock read?
(b) Later, the back of Anna’s ship passes Bob. At what time does this occur according to Bob?
(c) What will observers in Bob’s frame see on Anna’s two clocks at this time?
(d) Identify two events that show time dilation and two that show length contraction according to Anna.
The allowed electron energies predicted by the Bohr model of the hydrogen atom are correct.(a) Determine the three lowest. (b) The electron can "jump" from a higher to lower energy. with a photon carrying away the energy difference. From the three energies found in part (a), determine three possible wavelengths of light emitted by a hydrogen atom.
The density of copper is. Its femi energy is 7.0 eV, and it has one conduction electron per atom. At liquid nitrogen temperature (77 K), its resistivity is about . Estimate how far a conduction electron would travel before colliding and how many copper ions it would pass.
Exercise 81 obtained formulas for hydrogen like atoms in which the nucleus is not assumed infinite, as in the chapter, but is of mass, whileis the mass of the orbiting negative charge. (a) What percentage error is introduced in the hydrogen ground-state energy by assuming that the proton is of infinite mass? (b) Deuterium is a form of hydrogen in which a neutron joins the proton in the nucleus, making the nucleus twice as massive. Taking nuclear mass into account, by what percent do the ground-state energies of hydrogen and deuterium differ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.