Chapter 1: Q60E (page 1)
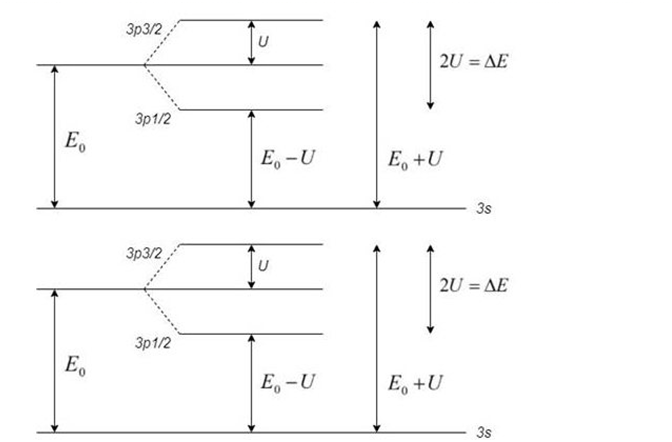
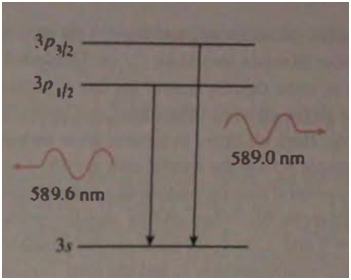
The well-known sodium doublet is two yellow spectral lines of very close wavelength.and It is caused by splitting of the energy level. due to the spin-orbit interaction. In its ground state, sodium's single valence electron is in the level. It may be excited to the next higher level. the 3p , then emit a photon as it drops back to the 3s . However. the 3p is actually two levels. in which Land Sare aligned and anti-aligned. (In the notation of Section these are. respectively. theand the because the (transitions Stan from slightly different initial energies yet have identical final energies(the having no orbital angular momentum to lead to spin-orbit interaction), there are two different wavelengths possible for the emitted photon. Calculate the difference in energy between the two photons. From this, obtain a rough value of the average strength of the internal magnetic field experienced by sodium's valence electron.
Short Answer
The magnetic field that the valence electron of sodium experiences is.