The electrons’ contribution to the heat capacity of a metal is small and goes to as . We might try to calculate it via the total internal energy, localid="1660131882505" , but it is one of those integrals impossible to do in closed form, and localid="1660131274621" is the culprit. Still, we can explain why the heat capacity should go to zero and obtain a rough value.
(a) Starting withexpressed as in equation (34), show that the slope atis.
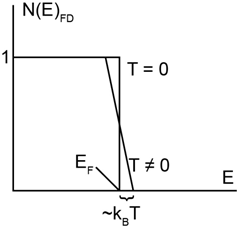
(b) Based on part (a), the accompanying figure is a good approximation to when is small. In a normal gas, such as air, whenis raised a little, all molecules, on average, gain a little energy, proportional to . Thus, the internal energy increases linearly with , and the heat capacity, , is roughly constant. Argue on the basis of the figure that in this fermion gas, as the temperature increases from to a small value , while some particles gain energy of roughly , not all do, and the number doing so is also roughly proportional to localid="1660131824460" . What effect does this have on the heat capacity?
(c)Viewing the total energy increase as simply = (number of particles whose energy increases) (energy change per particle) and assuming the density of states is simply a constant over the entire range of particle energies, show that the heat capacity under these lowest-temperature conditions should be proportional to . (Trying to be more precise is not really worthwhile, for the proportionality constant is subject to several corrections from effects we ignore).